Nuclear mechanics as a diagnostic and therapeutic target for glioblastoma
Glioblastomas (GBMs) are the most lethal primary brain tumours. The absence of effective therapies is mainly due to tumour invasion and to the resistance of invading cells to treatments such as radio and chemo-therapies. In GBMs, lamin proteins that control nuclear envelope stiffness, have recently emerged as potential markers of aggressiveness and tumourigenicity. Nuclear mechanics has appeared as a key determinant of cancer cell invasion leading us to hypothesize that genes controlling nuclear mechanics of GBM cells may be used as diagnostic tools and potential therapeutic targets to improve the prognostic of GBMs.
The working hypotheses of this M2 internship project is that alterations in nuclear mechanics contribute to GBM aggressiveness and directly influence cell invasive behaviour. The intern will first use clinically annotated primary patient-derived GBM cells and rheological techniques (optical tweezers, microfluidics) to measure nuclear morphology and mechanics (Figure). Second, he/she will modulate the expression levels of lamins to modify both nuclear mechanics and GBM cell invasion and test whether lamins could be used as potential molecular targets to control GBM aggressiveness.
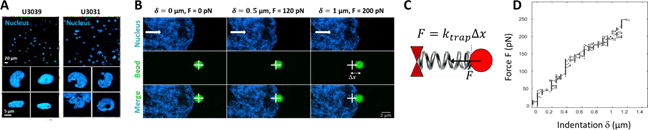
Figure: A. Comparison of the morphology of the nucleus in two different GBM cell lines (U3039 and U 3031). B. Measurements of the viscoelasticity of the nucleus using indentation of GBM nuclei in living cells. A. Images showing a typical nuclear indentation experiment. The white cross represents the centre of the optical tweezers in which the 2 µm- diameter bead is trapped (green). The nucleus (blue) is indented by moving the cell towards the right (white arrow) which displaces the bead away from the trap centre of a distance ∆x. C. Scheme of the bead in the optical trap. D. Force-indentation curve showing the force F as a function of the indentation d in the experiment shown in B.
Key words: nuclear envelope; lamin A/C; lamin B1; lamin B2; LINC complex; optical tweezers; microfluidic; cancer; glioblastoma; cytoskeleton; migration; invasion.
Collaborators: Sandrine Etienne-Manneville (Institut Pasteur, Paris), Catherine Villard (Institut Curie, Paris), Wang Xi (IJM, Paris), Nicolas Borghi (IJM, Paris)
Laboratory: Matière et Systèmes Complexes, UMR 7057 CNRS-Université de Paris, 10 Rue Alice Domon et Léonie Duquet, 75013 Paris
Contact: Jean-Baptiste Manneville (Jean-Baptiste.Manneville@u-paris.fr)
À lire aussi

Aquarium anéchoïque
Lorsque les parois d'une pièce réfléchissent trop le son, les réverbérations multiples peuvent vite rendre les conversations inintelligibles. Dans les salles de spectacle, les cinémas, et certaines salles de réunion, on cherche à traiter les surfaces, pour améliorer...

EvoDevMec2026 : Evolution, development and mechanics
We are happy to announce the forthcoming workshop entitled "Roadmap for EvoDevoMec", Nov. 2nd - 5th, 2026, Université Paris Cité. The link with evolutionary history of an organism is key to understand embryonic development. Much of the focus has been so far on genetic...

Le labo sur E=M6 !
Le démêlage des cheveux, une question de cosmétique, mais aussi de physique : c'est le sujet de la dernière émission du magazine "E=M6". Les journalistes sont venus à la rencontre des fabricants de brosse et de Nicolas Chevalier, chargé de recherche CNRS au...

Des icebergs motorisés
Les icebergs à la dérive sont principalement poussés par le vent, mais aussi par les courants océaniques et les vagues de surface. Dans le cadre d'une collaboration entre MSC, CNRS Paris Cité et Clark University aux Etats-Unis, Michael Berhanu, Amit Dawadi, Martin...
