Abstract below
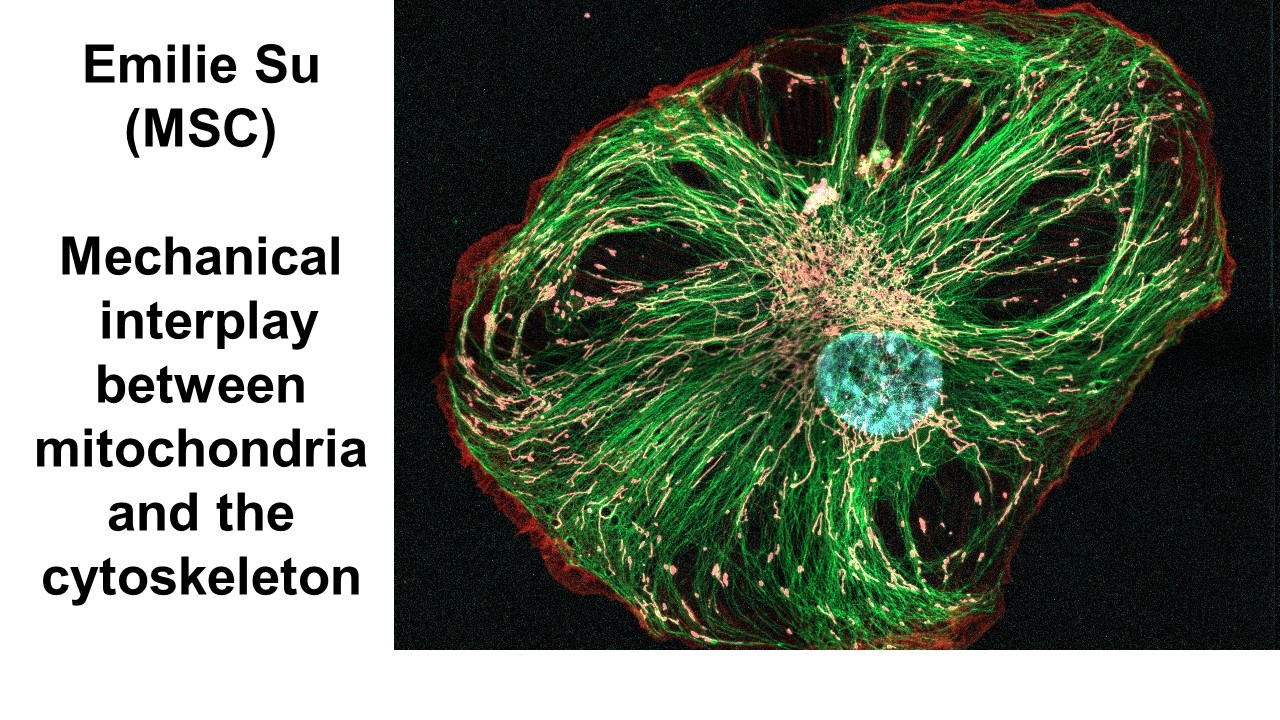
Emilie Su, MSC
PhD student under the supervision of Jean-Baptiste Manneville
Mechanical interplay between mitochondria and the cytoskeleton
Mitochondria are complex organelles involved in a wide range of cellular processes, including energy production and cell signalling. They thus sense and integrate both metabolic and mechanical cues to fine-tune their functions. Although their central role in energy metabolism has been extensively studied, the mechanical properties of mitochondria and their interactions with the cytoskeleton remain poorly understood.
Here we used an optical tweezers-based intracellular micromanipulation technique with endocytosed beads, to probe the mechanical properties of mitochondria and their microenvironment in RPE-1 cells. Our novel approach involves applying controlled forces to deflect single mitochondria, enabling the direct measurement of their effective stiffness while preserving their functional integrity. Additionally, we performed oscillatory intracellular rheology and selectively disrupted the actin and microtubule cytoskeletons to investigate their role in the mitochondrial microenvironment.
Our results reveal that the viscoelasticity of the cytoplasm is correlated with the local density of mitochondria only when actin is depolymerized and not in control conditions or when microtubules are disrupted. This indicates that actin filaments play a more significant mechanical role than microtubules in the mitochondrial microenvironment, at this spatio-temporal scale. Further experiments were conducted to assess the mechanical interplay between mitochondria and the cytoskeletal components, by quantifying force transmission through oscillations within the mitochondrial network. Our findings demonstrate that the actin cytoskeleton is the main mediator of force transmission to mitochondria.
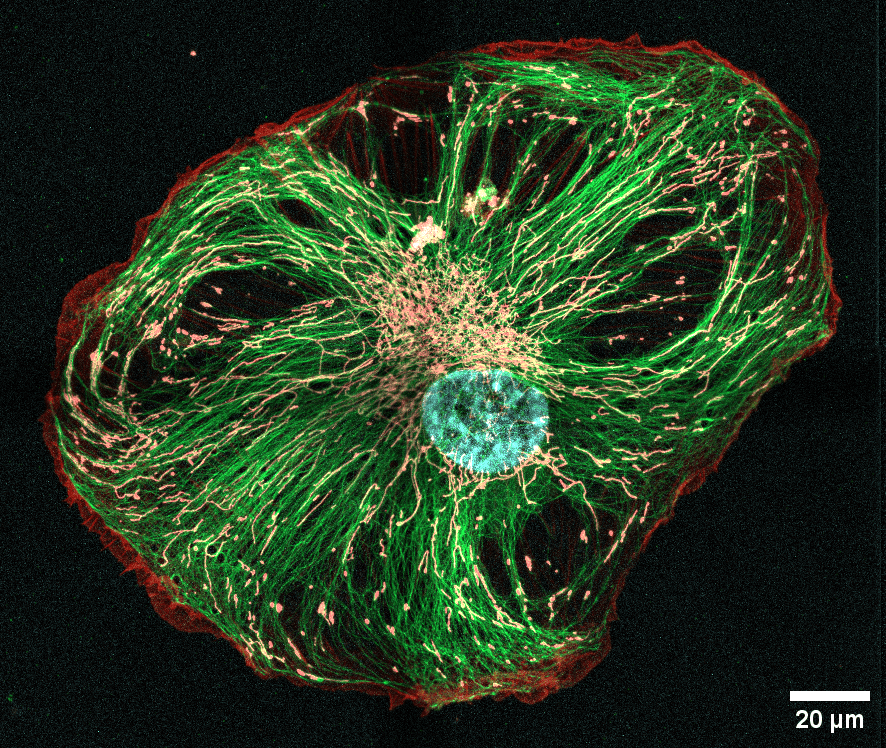
Legend: Confocal image of the mitochondrial and cytoskeletal networks (microtubules and actin) in a fixed RPE-1 cell.